- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:
ABSTRACT Metabolic rewiring has been recognized as a hallmark of malignant transformation, supplying the biosynthetic and energetic demands for rapid cancer cell proliferation and tumor
progression. A comprehensive understanding of the regulatory mechanisms governing these metabolic processes is still limited. Here, we identify the deubiquitinase ubiquitin-specific
peptidase 9 X-linked (USP9x) as a positive regulator of the proline biosynthesis pathway in non-small cell lung cancer (NSCLC). Our findings demonstrate USP9x directly stabilizes
pyrroline-5-carboxylate reductase 3 (PYCR3), a key enzyme in the proline cycle. Disruption of proline biosynthesis by either USP9x or PYCR3 knockdown influences the proline cycle leading to
a decreased activity of the connected pentose phosphate pathway and mitochondrial respiration. We show that USP9x is elevated in human cancer tissues and its suppression impairs NSCLC growth
in vitro and in vivo. Overall, our study uncovers a novel function of USP9x as a regulator of the proline biosynthesis pathway, which impacts lung cancer growth and progression, and
implicates a new potential therapeutic avenue. SIMILAR CONTENT BEING VIEWED BY OTHERS USP39 REGULATES PYRUVATE HANDLING IN NON-SMALL CELL LUNG CANCER Article Open access 18 December 2024
PINCH-1 REGULATES MITOCHONDRIAL DYNAMICS TO PROMOTE PROLINE SYNTHESIS AND TUMOR GROWTH Article Open access 01 October 2020 UBIQUITIN-SPECIFIC PROTEASE 54 REGULATES GLUT1-MEDIATED AEROBIC
GLYCOLYSIS TO INHIBIT LUNG ADENOCARCINOMA PROGRESSION BY MODIFYING P53 DEGRADATION Article 14 May 2024 INTRODUCTION Alterations in metabolic pathways promoting tumor growth represent a
hallmark of cancer [1]. Mechanistic insights into how the oncogenotype dictates metabolic patterns are well-understood and microenvironmental as well as genetic factors also appear to induce
a selective pressure that drives clonal evolution within tumors, which can generate or eliminate metabolic dependencies [2, 3]. However, we still possess a limited understanding of the
processes governing tumor metabolism to identify vulnerabilities suitable for an efficient therapeutic targeting in vivo. Lung cancer remains the leading cause of cancer-related deaths
worldwide [4] with non-small cell lung cancer (NSCLC) being the most common histological subtype. There is a great need for understanding the underlying pathomechanisms of NSCLC and for
defining disease-specific targets that would pave the way for the development of more effective therapies. During recent years, targeting metabolic addictions has been proposed as an
emerging approach [5]. In fact, it is well documented that lung cancer cells tend to shift their metabolic activities towards glycolysis, also known as the Warburg effect, to provide
precursors required for their rapid proliferation [6]. Increasing evidence further suggests that metabolic rearrangements in NSCLC not only occur in the pathways of central carbon
metabolism, but also in other conduits, such as in serine biosynthesis [7] or lipid metabolism [8]. Yet, defining a therapeutic window for targeting metabolic vulnerabilities differing
between proliferating cancer and normal cells remains challenging. Therefore, uncovering tumor-specific mechanisms of metabolic regulation emerges as a desirable promising therapeutic
avenue. Protein stability controlled by the ubiquitin proteasome system (UPS) can govern the abundance of proteins present in the cell by targeting polyubiquitinated proteins for recognition
and processing in the proteasome. Ubiquitin molecules are attached to a target protein by the enzymatic activities of E1 activating, E2 conjugating and E3 ubiquitin ligases and are
recognized by ubiquitin receptors [9]. In contrast, deubiquitinases (DUBs) have the opposing function of removing monoubiquitin or polyubiquitin chains from the target proteins [10]. The
balanced interplay of these two enzyme families coordinates ubiquitin tagging of proteins, ensuring a fine management of protein turnover, thereby maintaining optimally functioning proteins
[11]. Given their fundamental role in regulating protein stability and activity, it comes to no surprise that dysregulation of DUBs has been linked to most cancer-related pathways, both as
tumor promoters and tumor suppressors [12]. Numerous DUBs have been identified to directly contribute to cancer cell proliferation. For instance, OTUD7B deubiquitinates key cell cycle
regulators [13] and USP2 was shown to stabilize cyclin D1, a known proto-oncogene, causing an aberrant cell cycle progression in many different tumor types [14]. Furthermore, several DUBs,
including HAUSP/USP7, USP10, USP11, USP13, OTUD3, and Ataxin-3 have been shown to reverse the ubiquitination of PTEN, a key antagonist in the PI3K growth-promoting pathway, implicating its
function in cancer-specific contexts [15]. While exploration of therapeutic targeting of DUBs is an increasing area of research, most DUBs are poorly understood when it comes to their exact
function, targets, and regulation. Yet, due to their well-defined catalytic sites, DUBs represent suitable targets for small-molecule drug discovery [16]. Recently, stabilization of
metabolic enzymes through removal of ubiquitin has been proposed as one of the mechanisms by which cancer cells enhance their metabolic rate. Currently, only few DUBs out of the >90 DUBs
encoded in the human genome, including OTUB2, USP13, and JOSD2, have been attributed to display such function [17,18,19]. Whether additional DUBs can control protein stability relevant for
metabolic rewiring in cancer cells remains undiscovered. Here, we applied a systematic approach on all human DUBs in the context of lung cancer metabolism, starting with a broad in silico
analysis to narrow down to a DUB, ubiquitin-specific peptidase 9 X-linked (USP9x), with a potential role in regulating tumor growth via the proline metabolism pathway both in vitro and in
vivo. Our findings suggest a novel mechanism of proline biosynthesis regulation through direct stabilization of PYCR3 by USP9x. RESULTS DUBS ASSOCIATED WITH TUMOR METABOLISM ARE ELEVATED IN
HUMAN LUNG CANCER To identify DUBs that are potentially involved in tuning lung cancer metabolism, providing cells with elevated proliferation and growth ability, an unbiased analysis was
employed. We hypothesized that excessive DUB expression may be positively linked with growth-promoting metabolic pathways. Therefore, a guilt-by-association analysis on human DUB gene
expression data downloaded from The Cancer Genome Atlas (TCGA) public database (https://portal.gdc.cancer.gov/) was performed. All 99 DUBs were ranked according to similarity using Spearman
correlation followed by gene set enrichment analysis (GSEA) of the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways. This approach identified a cluster of 25 DUBs enriched for KEGG
terms associated with metabolic and growth-related activities, such as cell cycle, DNA replication, glycolysis, TCA cycle, pentose phosphate pathway, and arginine and proline synthesis (Fig.
1A), predicting that this set of DUBs may display a functional role in lung cancer metabolism and growth. Given that glycolysis is commonly dysregulated in lung cancer [20], it was examined
if the DUBs within the identified cluster displayed any effect on this metabolic pathway. Therefore, siRNA-mediated knockdown was performed for each of these DUBs and glycolysis was
measured in real-time in intact cells using an extracellular flux analyzer (Fig. 1B). Four DUBs (USP39, USP9x, OTUB1, and PSMD14) were identified, in which the knockdown resulted in a
significant reduction of glycolysis compared to the non-targeting siRNA control (_p_-values from 0.0005 – 0.0024). Notably, these four DUBs have been suggested to play roles in cancer
progression through promoting migration and metastasis [21,22,23,24]. However, as PSMD14 is a constituent of the 26 S proteasome, it is expected to impact general protein homeostasis
affecting metabolic pathways [25], OTUB1 was earlier reported to be directly linked to the regulation of the rate-limiting glycolytic enzyme, hexokinase 2 (HK2), [26], likely explaining the
effect observed by its knockdown on glycolysis. While USP39 is largely identified to participate in alternative splicing [27], the precise role of USP9x in different types of cancer, in
particular lung cancer, is less studied. To date, no direct metabolic enzymes have been identified as its targets. Therefore, we decided to continue our investigation on USP9x and its
potential role in cellular metabolism. To identify the metabolic pathways affected by the activity of USP9x, metabolite levels were measured using targeted capillary electrophoresis
time-of-flight/mass spectrometry (CE-TOF/MS) in USP9x-depleted lung cancer cells. Metabolite set enrichment analysis (MSEA) uncovered metabolic pathways altered specifically in the
USP9x-knockdown conditions compared to the control siRNA (Supplementary Table 1). The depletion of USP9x by two independent siRNAs led to significant changes in numerous pathways (Fig. 1C
and Supplementary Fig. 1A). Metabolic changes with at least 4 hits were observed in 9 pathways, primarily covering metabolites associated with amino acid metabolism, including glycine and
serine, arginine and proline, aspartate, alanine, glutamate, and the urea cycle. Collectively, these data suggest a potential role of USP9x as a positive regulator of metabolism in lung
cancer cells. USP9X REGULATES LUNG TUMOR GROWTH The guilt-by-association analysis in Fig. 1A predicted that USP9x, beyond being involved in metabolic pathways, was also associated with
growth-related activities. To investigate the impact of USP9x on tumor growth, in vitro and in vivo proliferation assays were performed by using doxycycline-inducible shRNA-expressing
NCI-H1975 lung cancer cell lines, targeting the USP9x gene or a non-targeting control (Fig. 2A). Using these cells, the clonogenicity and cell growth upon genetic silencing of USP9x in vitro
led to a significant growth reduction compared to the control (Fig. 2B, C). Furthermore, the impact of USP9x silencing on cell growth was measured in additional NSCLC cell lines, namely
A549 and NCI-H2087, revealing a similar growth reduction as in NCI-H1975 lung cancer cells (Fig. 2D). Importantly, the knockdown of USP9x did not induce cell death in either of the tested
lung cancer cell lines (Fig. 2E). To assess the in vivo effect of USP9x, inducible stable shRNA-expressing NCI-H1975 cell lines were subcutaneously injected into immunodeficient mice, which
had continuous access to doxycycline-containing drinking water to ensure constant shRNA expression and downregulation of USP9x. We found that genetic silencing of USP9x resulted in a drastic
reduction of tumor growth and tumor weight in vivo (Fig. 2F-H). These results argue that USP9x is critical for lung tumor growth, thus may be selectively elevated in lung cancer tissues
compared to non-neoplastic tissue. To test this hypothesis, we undertook two independent approaches. First, the gene expression data of lung adenocarcinoma (LUAD), the largest subgroup of
NSCLC, patients from the TCGA uncovered that USP9x transcripts were significantly enriched in lung tumors (_n_ = 535) compared to normal lung tissues (_n_ = 59; Fig. 2I). Secondly, to
evaluate USP9x protein levels in human lung cancer tissues, IHC staining of 142 LUAD samples of all disease stages was performed. By comparing the staining intensities to 92 normal adjacent
healthy tissues and normal lung tissues, our IHC analysis revealed that USP9x was specifically expressed in elevated levels in lung cancer tissues (Fig. 2J). While the normal lung tissues
displayed 64% of high expression of USP9x, the expression was elevated to 87% in the tumor samples. Taken together, these data suggest that USP9x is highly expressed in lung cancer tissues
and its expression promotes tumor growth. USP9X CONTROLS THE STABILITY OF PYCR3 DUBs target specific proteins and increase their stability through deubiquitination-mediated regulation.
Accumulation or simply the reduced clearance of these target proteins can lead to modulations of the pathway activities they are involved in. Some known specific targets for USP9x have been
previously identified and are linked to several cellular processes including cell cycle progression by inhibiting the degradation of Cdc20, a component of the mitotic checkpoint complex
[28], stress response by stabilizing apoptosis signal-regulating kinase 1 (ASK1) [29], or chromosome alignment by deubiquitinating Survivin [30]. To identify possible metabolic enzymes
directly targeted by USP9x, co-immunoprecipitation experiments were performed to elucidate the interaction partners of USP9x. Mass spectrometric analysis of the co-immunoprecipitated
proteins revealed several potential interaction partners specific to USP9x (Supplementary Table 2). By focusing on metabolism-regulating enzymes among the identified hits, the
pyrroline-5-carboxylate reductase 3 (PYCR3) was identified as a potential interaction partner of USP9x with 17 unique peptides detected, which covered 43% of the protein sequence (Fig. 3A).
Immunoblotting further confirmed the presence of this interaction (Fig. 3B). To test the impact of USP9x on PYCR3 deubiquitination, the deubiquitination activity of USP9x was assessed by
treating cells with the proteasomal inhibitor MG132, which prevents the degradation of ubiquitinated proteins. Overexpression of USP9x markedly reduced the level of ubiquitinated PYCR3
compared to the control (Fig. 3C), suggesting that PYCR3 is directly deubiquitinated by USP9x. Next, we examined whether USP9x knockdown or overexpression augments the stability of PYCR3.
While both the depletion of USP9x by two independent siRNAs and its inhibition with the selective USP9x inhibitor FT709 reduced PYCR3 protein levels (Fig. 3D, E), USP9x overexpression
resulted in elevated PYCR3 protein levels (Fig. 3F). Since knockdown of USP9x did not influence PYCR3 transcription (Fig. 3G), we concluded that PYCR3 is directly regulated by USP9x on the
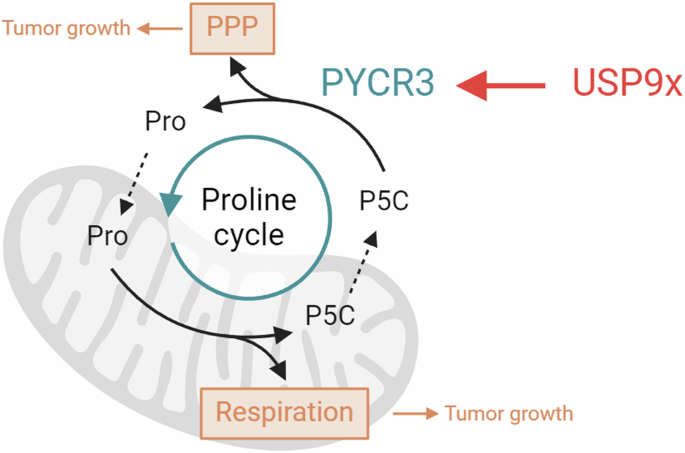
protein level. Hence, we identified PYCR3 as a novel target of USP9x. USP9X ALTERS THE PROLINE METABOLISM PATHWAY There are three human PYCR isoforms all known to play key roles within
proline biosynthesis. The PYCR3 isoform catalyzes the reaction from pyrroline-5-carboxylate (P5C) to proline (Fig. 4A) in the cytosol by utilizing NADPH as a cofactor [31]. In the
PYCR3-mediated branch of proline biosynthesis, P5C mainly originates from arginine, ornithine and therefore the urea cycle, while glutamate from the TCA cycle can serve as an alternative
source. Our unbiased MSEA of metabolomics data from USP9x depleted cells (Fig. 1C) revealed arginine and proline metabolism among the most significantly altered pathways. Therefore, we
focused on individual metabolite levels within the proline biosynthesis pathway to investigate the potential role of USP9x in regulating proline biosynthesis through its effect on the PYCR3
enzyme. Depletion in USP9x led to an accumulation of arginine and ornithine, which are the preferred sources of the PYCR3-mediated proline pathway (Fig. 4B). Minor changes were observed in
the alternative sources for the PYCR3-mediated route, namely α-ketoglutarate and glutamate (Supplementary Fig. 1B). Furthermore, proline, the product the proline biosynthesis pathway, was
not found to be altered upon USP9x knockdown (Fig. 4B), which is mainly synthesized by PYCR1/2 in the mitochondria. This observation is in line with previous observations that PYCR3 mainly
contributes to the proline cycle and not to the overall proline pool within the cell [31]. Cellular fractionation further confirmed that USP9x and PYCR3 are both localized in the cytosol
(Fig. 4C). Combined, these results reveal a role for USP9x in the PYCR3-mediated branch of the proline biosynthesis pathway through its regulation of PYCR3. The proline cycle acts as a redox
shuttle between the cytosol and mitochondria transferring NAD + /NADH and NADP + /NADPH to maintain redox homeostasis. It has been reported that recycling of NADP + /NADPH connects proline
biosynthesis with the oxidative phase of the pentose phosphate pathway (PPP), contributing to the synthesis of nucleotides [32]. Therefore, we investigated the impact of USP9x depletion on
PPP to assess whether its targeting of PYCR3 affects the proline cycle. Indeed, both G6PDH activity and the 6-phosphogluconate/glucose-6-phosphate metabolite ratios were significantly
reduced upon genetic silencing of USP9x (Fig. 4D, E), indicating a reduced activity of the NADPH-dependent reactions of the PPP. Furthermore, the ratio of NADP + /NADPH was reduced,
suggesting the accumulation of NADPH due to lowered PYCR3 activity upon USP9x knockdown (Fig. 4F). Accordingly, we observed dysregulation of the PPP as well as NADP + /NADPH homeostasis upon
USP9x depletion, strengthening our conclusion of USP9x targeting PYCR3 and thus the proline cycle. In the mitochondrial segment of the proline cycle, proline is degraded to P5C, a reaction
catalyzed by proline dehydrogenase (PRODH). In this reaction, PRODH transfers electrons through FADH2 to ubiquinone, which is part of the electron transport chain [33]. Therefore, we
investigated whether dysregulation of PYCR3 might also affect mitochondrial respiration. Indeed, knockdown of PYCR3 (Fig. 4G) resulted in a significant reduction of mitochondrial respiration
(Fig. 4H, I). Moreover, depletion of PYCR3 displayed cell growth retardation in vitro (Fig. 4J). These results suggest a regulatory role of PYCR3 in lung cancer cell growth, likely
dependent on its involvement in the proline cycle and its impact on related growth-promoting pathways. We further investigated the potential clinical relevance by examining survival data of
lung cancer patients with PYCR3-high _versus_ PYCR3-low expression levels. The Kaplan–Meier analysis showed that high expression levels of PYCR3 significantly (_p_-value 0.037) predicted
poor prognosis (Fig. 4K). Collectively, these findings suggest that USP9x regulates the proline biosynthesis pathway and the proline cycle via PYCR3, which affects PPP and mitochondrial
respiration. DISCUSSION Cancer cells rewire metabolic pathways to supply their energetic and biosynthetic demands for tumorigenesis and rapid proliferation [6]. These metabolic
rearrangements are complex and still under investigation. Here, we uncovered a novel aspect of metabolic regulation in lung cancer cells that could offer new avenues for therapeutic
interventions. DUBs are important contributors of healthy protein homeostasis, and their dysregulation has been linked to multiple human diseases highlighting their critical physiological
role [16]. Despite many recent efforts, the involvement of DUBs in managing the activity of metabolic pathways in cancer cells is still poorly understood. The deubiquitinase USP9x has been
previously studied in several types of cancer including its involvement in the promotion of liver cancer through modulation of JAK2/STAT3 signaling [34]. In a study on breast cancer, USP9x
was found to promote chemoresistance through the stabilization of the transcriptional regulator YAP1 [35]. However, to date, the only known link between USP9x and cellular metabolism has
been the fact that USP9x can stabilize mTOR kinase complexes [36, 37], but no direct metabolic target has been identified yet. In this study, we present that USP9x interacts with the PYCR3
enzyme, promoting its stability through deubiquitination-mediated regulation, thus revealing a previously uncovered role of this DUB as a direct regulator of proline metabolism in cancer
cells. The disruption of the proline cycle interfered with redox homeostasis and thereby with the PPP and mitochondrial respiration, which are critical for cancer growth. While the proline
cycle has been suggested as a potential therapeutic target in cancer [38], very little is known about its regulation. Kindlin-2 has been shown to be a binding partner of PYCR1 leading to its
stabilization in response to the cell mechano-environment [39]. Whether this has implications on the proline cycle or whether Kindlin-2 also targets PYCR3 remains to be further
investigated. To our knowledge, no studies have been published yet suggesting how PYCR3 is regulated in cancer so far. Our study demonstrates USP9x as a direct interactor of PYCR3 preserving
enhanced PYCR3 stability. As a result, USP9x activity allows to maintain proline cycling in cancer cells. Given that USP9x depletion significantly affected metabolism in NSCLC cells,
warrants this DUB as an important regulator of cancer metabolism. Collectively, our findings provide a novel understanding of the governance of the proline biosynthesis pathway, specifically
the PYCR3-mediated branch, in lung cancer cells. Treatment approaches targeting metabolic vulnerabilities in NSCLC are complicated due to metabolic heterogeneity in cancer patients.
Understanding which metabolic profiles exist in lung cancer patients and how these profiles are shaped is important for improving the treatment success. We have previously shown that the DUB
JOSD2 is involved in maintenance of serine metabolism in NSCLC and that enhanced serine pathway activity is linked with poor prognosis [40]. Now, our current findings solidify DUBs as
important regulators of metabolic rewiring in cancer, warranting DUBs as promising targets for therapeutic intervention of tumor metabolism. MATERIALS AND METHODS CELL CULTURE The NSCLC cell
lines A549, NCI-H1975, and NCI-H2087 were grown in RPMI-1640 medium (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 10% heat-inactivated FBS (Gibco, Waltham, MA, USA), 2 mM
L-glutamine (Sigma-Aldrich), and 100 U/mL penicillin/streptomycin (Sigma-Aldrich). For culturing of the stable Tet-inducible shRNA knockdown cell lines, the medium contained 10%
Tet-system-approved FBS (Gibco). The human embryonic kidney cell line HEK293T was cultured in DMEM medium (Gibco) supplemented with 10% heat-inactivated FBS, 2 mM L-glutamine, 100 U/mL
penicillin/streptomycin, as well as 1% non-essential amino acids (Sigma-Aldrich), and 100 µM sodium pyruvate (Sigma-Aldrich). All cell lines were kept in a humidified incubator at 37 °C and
5% CO2 in a logarithmic growth phase. Frequent PCR-based testing verified the absence of mycoplasma contamination [41, 42]. ANALYSIS OF PRIMARY NSCLC PATIENT DATA For the
guilt-by-association analysis, Spearman correlation was performed between the levels of each of the 99 DUBs used in the screen and the levels of all transcripts, which was computed across
all LUAD samples from the TCGA data portal (https://portal.gdc.cancer.gov/). Based on the correlations, the genes were ranked, and used as input for the GSEA analysis of the KEGG pathways
using the clusterProfiler package in R [43]. We colored the corresponding DUB-pathway blocks red for highly correlated genes and blue for highly anti-correlated genes, with the color scheme
based on the log10(_p_-value). The RNA expression levels of USP9x in LUAD tissues (_n_ = 535) were compared to normal lung tissues (_n_ = 59). The data was downloaded from the TCGA data
portal and visualized as violin plots. Overall survival of lung cancer patients (_n_ = 504) was analyzed by Kaplan–Meier plot with data downloaded from https://kmplot.com/ and based on the
RNA expression data from the TCGA. The cutoff to divide patients into PYCR3 expression high and low groups was determined by the performance of all possible cutoff values between the lower
and upper quantiles. Plots were generated using the survminer package in R [44]. GLYCOLYSIS SCREEN Cells were transfected with siRNAs targeting the following DUBs: USP15, FBXO8, USP14, UCK2,
USP18, UBL4A, UBL5, STAMBP, UDF1L, UCHL3, COP3S, UCHL5, UCHL1, OTUB2, SENP2, USP1, BRCC3, OTUD6B, UEVLD, USP5, USP10, OTUB1, PSMD14, USP39, and USP9x (ON-TARGETplusTM siRNA pools purchased
from Dharmacon, Lafayette, CO, USA). Cells were reseeded into XFe96 cell culture miniplates (6 000 cells/well) 48 h post transfection. On the next day, they were washed and supplied with XF
Base medium (Seahorse Bioscience, North Billerica, MA, USA) containing 2 mM L-glutamine, left at 37 °C for 20 min, and then subjected to the assay in an XFe96 Extracellular Flux Analyzer
(Seahorse Bioscience). After three baseline measurements, 10 mM glucose and 50 mM 2-deoxyglucose were injected to the cells and three ECAR (extracellular acidification rate) measurements
were done after each injection. The obtained ECAR levels were normalized to the protein concentration of each well as measured by BCA assay. Data were plotted as difference in ECAR between
DUB knockdown and non-targeting siRNA control. Target Product No. Target Product No. BRCC3 L-005798-00 UCHL5 L-006060-00 COPS5 L-005814-00 UCK2 L-005077-00 FBXO8 L-012435-00 UDF1L
L-017918-00 OTUB1 L-021061-00 UEVLD L-008494-02 OTUB2 L-010983-00 USP1 L-006061-00 OTUD6B L-008553-00 USP10 L-006062-00 PSMD14 L-006024-00 USP14 L-006065-00 SENP2 L-006033-00 USP15
L-006066-00 STAMBP L-012202-00 USP18 L-004236-00 UBL4A L-020658-00 USP39 L-006087-00 UBL5 L-014320-00 USP5 L-006095-00 UCHL1 L-004309-00 USP9x L-006099-00 UCHL3 L-006059-00 GENERATION OF
INDUCIBLE SHRNA-EXPRESSING CELL LINES For the generation of inducible shRNA-expressing cell lines, shRNA oligomers targeting USP9x (shRNA 1: GAGAGTTTATTCACTGTCTTA, shRNA 2:
CGCCTGATTCTTCCAATGAAA) were cloned into the Tet-pLKO-puro plasmid (gift from Dmitri Wiederschain (Addgene plasmid #21915 ; http://n2t.net/addgene:21915 ; RRID:Addgene_21915)) as previously
described [45, 46]. A plasmid with a non-targeting randomized shRNA sequence was used for control (gift from Roland Friedel (Addgene plasmid #98398; http://n2t.net/addgene:98398;
RRID:Addgene_98398), [47]). Lentiviral particles were produced in HEK293T cells by co-transfecting the cloned shRNA constructs, psPAX2 (gift from Didier Trono (Addgene plasmid #12260;
http://n2t.net/addgene:12260; RRID:Addgene_12260)), and pMD2.G (gift from Didier Trono (Addgene plasmid #12259 ; http://n2t.net/addgene:12259 ; RRID:Addgene_12259)) in a 2.5:1.5:1 ratio. The
plasmid mixtures were transfected using LipofectamineTM 2000 (Invitrogen, Thermo Fisher, Waltham, MA, USA) per the manufacturer’s protocol. Medium containing lentiviral particles was
collected after 48 and 72 h. NCI-H1975 cells were transduced with the lentivirus-containing medium in the presence of 12 µg/mL polybrene (Sigma-Aldrich) for 24 h. Stable cell lines were
generated by selection with 1.5 µg/mL puromycin (Sigma-Aldrich) for 14 days. Knockdown was induced with 20 ng/mL doxycycline (Sigma-Aldrich) for 48 h. SIRNA-MEDIATED KNOCKDOWN Cells were
forward transfected using either the Interferin transfection reagent (Polyplus transfection, New York, NY, USA) or the Lipofectamine™ RNAiMAX transfection reagent (Invitrogen, Thermo Fisher)
following the recommended manufacturers’ protocols. A non-targeting siRNA served as a control. Knockdown efficiencies were confirmed 48 h post transfection by qPCR or western blot. Target
Company Product No NT Control Pool Dharmacon, Lafayette, CO, USA D-001810-10 USP9x siRNA #1 Dharmacon, Lafayette, CO, USA J-006099-06 siRNA #2 Dharmacon, Lafayette, CO, USA J-006099-08 PYCR3
Dharmacon, Lafayette, CO, USA L-014246-00 PHARMACOLOGICAL INHIBITION OF USP9X FT709, a specific inhibitor of USP9x [48] was purchased from MedChemExpress (HY-145967, Monmouth Junction, NJ,
USA) and prepared according to the manufacturer’s instructions. At approximately 80% confluency, cells were treated with 20 µM FT709 or DMSO only control for 48 h, then collected, lysed and
PYCR3 levels were assessed by western blotting. PLASMID TRANSFECTION A USP9x-coding plasmid vector (RC217531) was purchased from Origene. At 80% confluency, the cells were transfected in
6-well plates with either pCMV empty vector or USP9x plasmid vector using the ViaFect transfection reagent (Promega, Madison, WI, USA) following the manufacturer’s protocol. The transfection
mix contained 100 µL of Opti-MEM medium, 2 µg of the plasmid vector and 4 µL of ViaFect per 1 mL of cell culture medium. At 24 and 48 h after transfection, the cells were harvested and
lysed for western blotting. WESTERN BLOT Cells were lysed using the cOmplete™ Lysis-M buffer (Roche Applied Science, Mannheim, Germany) with protease inhibitors (Roche Diagnostics,
Risch-Rotkreutz, Switzerland) and the protein concentrations determined using the Pierce BCA Protein Assay kit (Thermo Fisher Scientific). Samples were mixed with Laemmli sample buffer
(BioRad, Hercules, CA, USA), heated, and loaded in equal amounts onto 10% SDS-PAGE. After transfer onto nitrocellulose membranes, they were blocked using 5% non-fat dairy milk in PBS and
0.1% TWEEN® 20 at room temperature. Subsequently, membranes were probed with primary antibodies (diluted in PBS with 1% BSA and 0.1% NaN3) overnight at 4 °C. Secondary HRP-conjugated
antibodies (goat anti-mouse or goat anti-rabbit, Thermo Fisher Scientific) were diluted in PBS with 2.5% non-fat dairy milk and 0.05% TWEEN® 20. Finally, membranes were developed using
Clarity TM Western ECL (BioRad). Protein levels were quantified using the ImageJ software. Target Company Product no. β-Actin Santa Cruz Biotech, Dallas, TX, USA sc-81178 PYCR3 ProteinTech
Group, Chicago, IL, USA 68087-1-Ig TOMM40 Santa Cruz Biotech, Dallas, TX, USA sc-11414 Tubulin Sigma-Aldrich, St. Louis, MO, USA T8203 Ubiquitin Cell Signaling Technology, Danvers, MS, USA
3933 S USP9x ProteinTech Group, Chicago, IL, USA 55054-1-AP Vinculin Abcam, Cambridge, UK ab129002 QPCR RNA was extracted using the PureLink™ RNA Mini Kit (Invitrogen, Thermo Fisher
Scientific) and treated with DNase (Thermo Fisher Scientific). For cDNA synthesis, 1 µg of RNA was transcribed using the iScript cDNA synthesis kit (BioRad). The qPCR was performed with cDNA
corresponding to 10 ng of total RNA and the Maxima qPCR SYBR green master mix (Thermo Fisher Scientific) and run on the StepOnePlus™ Real-Time PCR System (Applied Biosystems, Waltham, MA,
USA). The relative mRNA expression was calculated with the ΔΔCT method, Tubulin was used as the housekeeping gene. Target Sequence PYCR3 fw 5’-GCCCCCAAACACACGGG-3’ rv
5’-CCATCACTATGGCCCCTTCCT-3’ Tubulin fw 5’-TCTACCTCCCTCACTCAGCT-3’ rv 5’-CCAGAGTCAGGGGTGTTCAT-3’ USP9x fw 5’-GTGTCAGTTCGTCTTGCTCAGC-3’ rv 5’-GCTGTAACGACCCACATCCTGA-3’ IN VITRO GROWTH ASSAYS
After inducing the knockdown with either doxycycline in the stable shRNA-expressing cell lines or performing the transfection with siRNA, cells were reseeded in triplicates in a 96-well
plate at 1 000 – 2 000 cells/well. Cell growth was assessed for 4 days using the CellTiter-Glo® luminescent cell viability assay (Promega, Madison, WI, USA). The signal was read on a GloMax
Discover Microplate Reader (Promega) and the values normalized to the 0 h time point and to the respective controls. For the clonogenic growth assay, cells were reseeded after induction of
the knockdown with doxycycline for 48 h at 1 000 cells/well in 12-well plates. Medium was replaced every 72–96 h and the cells cultured for a total of 7–9 days. Cells were then washed with
PBS, fixed with 1% paraformaldehyde (Sigma-Aldrich), and stained with 0.1% crystal violet (Sigma-Aldrich). For quantification, each well was treated with 200 µL 1 M NaOH, 2 × 75 µL
transferred into a black 96-well plate, and the solution neutralized with 1 M HCl. Fluorescence was detected using the GloMax Discover Microplate Reader, with an excitation wavelength of 520
nm and emission wavelength of 580–640 nm. The background fluorescence was subtracted, and the values normalized to the control condition. CELL TOXICITY Cell toxicity after siRNA-mediated
knockdown was determined using the bioluminescent ToxiLightTM BioAssay Kit (Lonza, Basel, Switzerland) using the manufacturer’s protocol. Cell toxicity was measured after 48 and 96 h post
transfection and the values normalized to the former time point. ANIMAL STUDIES For the in vivo studies, female 4–5 week old immunodeficient Athymic nude mice (Rj:ATHYM-Foxn1nu/nu*) were
purchased from Janvier Labs (Le Genest-Saint-Isle, France). Mice were housed in pathogen-free conditions at the Karolinska Institute animal facility with temperatures between 20–24 °C and a
12 h light/dark cycle. All experiments were performed in accordance with the Swedish animal welfare laws authorized by the Stockholm Animal Ethics Committee. For the tumor growth study, 18
mice were randomly divided into 3 groups, each group consisting of 6 mice prior to injection of shRNA-expressing cells. IN VIVO TUMOR GROWTH Stable shRNA-expressing NCI-H1975 cells were
treated with 20 ng/mL doxycycline for 48 h to ensure homogenous induction of the knockdown prior the subcutaneous injection of 3 × 106 cells/mouse in 100 µL of 1:1 PBS and Matrigel (Corning
Inc., Corning, NY, USA) into the flanks. During the experiment, mice were provided with doxycycline-containing drinking water (2 mg/mL doxycycline, supplemented with 5% sucrose
(Sigma-Aldrich)). To track the tumor volume, tumors were measured with a caliper every third day. At the end of the experiment, the mice were sacrificed, the tumors were removed, weighed,
frozen in liquid nitrogen, and stored at -80 °C until further analysis. To calculate the tumor volume, the following formula was used: 2 W x L/2 (L = length and W = the perpendicular width
of the tumor, L > W). The knockdown efficiency of the tumors was validated by qPCR. IMMUNOHISTOCHEMISTRY Immunohistochemistry staining for USP9x was performed using the Autostainer 480
(Thermo Fisher Scientific, Waltham, MA, USA) as previously described [49]. Briefly, tissue slides were incubated in Ultra V block (Thermo Fisher Scientific) for 5 min and stained with an
antibody targeting USP9x (CAB070164, Human Protein Atlas) for 30 min. Afterwards, slides were incubated with HRP polymer for 30 min, with 3,3′-diaminobenzidine solution for 5 min,
counterstained in Mayers hematoxylin for 5 min using the Autostainer XL (Leica Biosystems, Nussloch, Germany), and finally they were rinsed in lithium carbonate water for 1 min. The slides
were dehydrated in graded ethanol and coverslipped (PERTEX, Histolab) using the CV5030 automated glass coverslipper (Leica Biosystems, Nussloch, Germany). The slides were scanned using the
Aperio XT automated scanning system (Leica Biosystems, Nussloch, Germany). The non-neoplastic tissues included normal lung, adjacent lung, and matched adjacent lung tissues (_n_ = 92). All
tumor tissues were lung adenocarcinoma (_n_ = 142). The following scoring criteria were used for the immunohistochemical stainings as analyzed with ImageJ; negative (0), weak (1), moderate
(2), and strong [3]. USP9x-high includes strong (3) and USP9x-low includes (2) down to (0). CO-IMMUNOPRECIPITATION For co-immunoprecipitation experiments, NCI-H1975 cells were seeded in 150
mm2 dishes (3 dishes per condition) and collected the next day by scraping. The cells were pelleted by centrifugation and lysed in lysis buffer from the MS/MS kit (Pierce™ MS-Compatible
Magnetic IP Kit, Thermo Fisher) for 10 min on ice. After centrifugation (13 000 _g_, 5 min), the supernatant was collected, the protein concentration was measured, and the samples equally
divided into several tubes including isotype antibody control (Abctr5 Proteinlab or Abctr6 Thermo Fisher) and USP9x. 20 µL of the Protein A/G Magnetic Beads (Pierce™ MS-Compatible Magnetic
IP Kit, Thermo Fisher) were conjugated with either the isotype antibody control or the USP9x antibody (1.5 µg each) in lysis buffer for 6 h at 4 °C on a rotating platform. Then, the
antibody-bead complexes were washed once with the lysis buffer and incubated overnight with the cell lysates. The beads were washed according to the kit protocol. 10% of the bead-containing
protein complexes was used for immunoblotting and the remaining part (corresponding to 0.6 mg of initial total protein lysate) was sent for the LC-MS/MS analysis. MASS SPECTROMETRIC ANALYSIS
OF IMMUNOCONJUGATES Mass spectrometric analysis was performed by the Proteomics Biomedicum Core Facility at the Karolinska Institute. Immunoconjugates were digested with trypsin with the
complexes remaining on the magnetic beads. Peptides were purified and resuspended in 2% acetonitrile (ACN), 0.1% formic acid (FA) in a total volume of 15 µL. For the Nano-flow LC-MS/MS
analysis, 2 µL of the samples were injected into the Ultimate 3000 UHPLC chromatographic system (Thermo Fisher Scientific) coupled on-line to a Q Exactive™ HF hybrid Quadrupole-Orbitrap™
mass spectrometer (Thermo Fisher Scientific). Separation of the peptides was done on a 50 cm long heated (55 °C) C-18 Easy-Spray™ column (Thermo Fisher Scientific) using an organic gradient
4–36% B in 120 min at 300 nL/min flow rate (solvent A: 2% ACN, 0.1% FA; solvent B: 98% ACN, 0.1% FA). Survey mass spectra were acquired at mass resolution of 120 000 (range of m/z 350-600).
MS/MS data of the 17 most intensive precursors were obtained at a resolution of 30 000 by higher-energy collisional dissociation (HCD) with 28% normalized collision energy. The in-house
Raw2mgf program was used to convert the MS raw data files into Mascot Generic Format (mgf). The mgf files were searched against the SwissProt database (HUMAN) with the Mascot Server search
engine (v2.5.1, MatrixScience Ltd., London, UK) allowing up to two missed cleavage sites for trypsin, a mass tolerance of 10 ppm, and 0.02 Da for the precursors and HCD fragment ions,
respectively. Cysteine carbamidoethylation was used as fixed modification, while deaminations of asparagine and glutamine, as well as oxidation of methionine were dynamic. Proteins with any
number of detected unique peptides in the USP9x conjugates and no peptides in both controls were considered as potential interaction partners. UBIQUITINATION ASSAY NCI-H1975 cells were
co-transfected with overexpression plasmids using the above-described procedure and using the following constructs: Control Vector (pCB6 + )/USP9x overexpression plasmid (RC217531, Origene)
+ PYCR3 overexpression plasmid (RC203382, Origene) + HA-Ubiquitin overexpression vector (gift from Edward Yeh (Addgene plasmid #18712; http://n2t.net/addgene:18712; RRID:Addgene_18712),
[50]). At 24 h later, cells were treated with 5 µM of the proteasomal inhibitor MG132 (Selleckchem, Houston, TX, USA) for 4 h prior harvesting. The lysates were used for immunoprecipitation
with the antibody against PYCR3. Proteins were resolved by western blotting on a 12% SDS-PAGE. Immunoblotting with anti-Ubiquitin and anti-PYCR3 antibodies was performed to assess the
ubiquitination status PYCR3 upon USP9x overexpression. Level of ubiquitinated PYCR3 was quantified using ImageJ. METABOLOMICS At 48 h post transfection with knockdown-inducing siRNAs,
metabolites were extracted from the cells as previously described [51]. Briefly, cells were washed with 5% mannitol (Sigma-Aldrich) solution and metabolites extracted with methanol
(Sigma-Aldrich) containing 10 µM Internal Standard Solution (Human Metabolome Technologies, Boston, MA, USA). The extracted metabolites were centrifuged at 2 300 _g_ at 4 °C for 5 min and
the supernatant filtered through a Millipore 5-kDa cutoff filter at 9 100 _g_ at 4 °C for 2 h. The filtrate was analyzed using Capillary-Electrophoresis Time of Flight Mass Spectrometer
(CE-TOF/MS, Human Metabolome Technologies). METABOLITE SET ENRICHMENT ANALYSIS (MSEA) For the metabolite set enrichment analysis (MSEA) the MetaboAnalystR package was used. Dot plots were
used to represent the pathways that were significantly impacted by knockdown of both siRNAs, with the color and size of the dots representing the -log10 of the corrected _p_-value and the
number of metabolites that were enriched in each pathway, respectively. SUB-CELLULAR FRACTIONATION The NCI-H1975 cells were seeded on 100 mm2 dishes one day before the experiment. The cells
were washed twice with PBS and collected using cell scrapers. The cell suspension was equally divided among two tubes to prepare total lysate and cellular fractions. The total lysates were
prepared by lysing cells for 15 min on ice in RIPA buffer, supplemented with cOmpleteTM protease and phosphatase inhibitor cocktails. The cytosolic fraction was prepared by lysing cells in
buffer A (150 mM NaCl, 50 mM Hepes, pH 7.4, 0.02% digitonin, cOmpleteTM protease and phosphatase inhibitor cocktails) and incubated for 5 min at room temperature. The obtained extract was
centrifuged at 15 000 _g_ for 10 min and the supernatant was collected as the cytosolic fraction. Then, the pellets were washed 3 times with PBS and the membrane fraction containing
mitochondria was extracted using buffer B (RIPA lysis buffer supplemented with cOmpleteTM protease and phosphatase inhibitor cocktails) for 15 min on ice. After centrifugation (15 000 _g_,
10 min), the supernatant was collected as the fraction containing mitochondria. TOMM40 served as a marker for the membrane fraction, and Tubulin for the cytosolic fraction. G6PDH ACTIVITY
ASSAY The G6PDH enzymatic activity was determined using the colorimetric assay kit by Abcam (ab102529) and following the manufacturer’s instructions. Briefly, cells were harvested 48 h post
transfection with siRNAs for USP9x. After centrifugation at 12 000 _g_ for 5 min at 4 °C, the supernatant was collected for subsequent analysis. The reaction mix was added according to the
protocol and the absorbance measured after 6 min at 450 nm wavelength on the GloMax Discover Microplate Reader (Promega) at 37 °C. The enzymatic activities were normalized to protein
concentrations and to the control condition. NADP + /NADPH ASSAY NADP+ and NADPH levels were determined using the bioluminescent NADP/NADPH-GloTM Assay by Promega (G9081). Briefly, cells
were lysed in base solution with 1% DTAB (Sigma-Aldrich). The cell lysates were split into two parts to enrich for NADP+ and NADPH by acid and base treatment, respectively. To measure NADP +
, the samples were treated with 0.4 N HCl and heat quenched at 60 °C for 15 min, while the NADPH samples were heat quenched in the 1% DTAB base solution. The samples were neutralized with
Trizma or HCl-Trizma after cooling down to room temperature. The treated cell lysates were mixed in equal parts with the NADP/NADPH-GloTM detection reagent, incubated for 45 min, and the
luminescence measured with the GloMax Discover Microplate Reader. DETERMINATION OF MITOCHONDRIAL RESPIRATION To determine mitochondrial respiration, the oxygen consumption rate (OCR) was
measured in real-time using the XFp Extracellular Flux Analyzer (Seahorse Bioscience). After induction of the knockdown using siRNA transfection for 48 h, cells were reseeded in triplicates
at a density of 6 000 cells/well in an XFp miniplate. On the next day, cells were washed, and the medium changed to XF Base medium (Seahorse Bioscience) with 1 mM pyruvate, 2 mM glutamine,
and 10 mM glucose about 30 min before the start of the assay. After a baseline measurement, 1 μM oligomycin, 0.5 µM FCCP, and 0.5 µM rotenone/antimycin A were injected to the cells over the
course of approximately 1.5 h. STATISTICAL ANALYSIS All graphs were designed, and the statistics calculated using the GraphPad Prism Software version 9.0.0 (Dotmatics, Boston, MA, USA) or R
version 4.2.0. The data were presented as the mean ± S.D. Unless otherwise mentioned, unpaired _t_-tests were used for statistical analysis. The specifics for each panel are described in the
respective figure legend. *_p_ ≤ 0.05, **_p_ ≤ 0.01, ***_p_ ≤ 0.001, ****_p_ ≤ 0.0001. DATA AVAILABILITY The datasets generated during and/or analyzed during the current study are available
from the corresponding author on reasonable request. REFERENCES * Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74. Article CAS PubMed Google
Scholar * Stanley IA, Ribeiro SM, Gimenez-Cassina A, Norberg E, Danial NN. Changing appetites: the adaptive advantages of fuel choice. Trends Cell Biol. 2014;24:118–27. Article CAS PubMed
Google Scholar * Stine ZE, Schug ZT, Salvino JM, Dang CV. Targeting cancer metabolism in the era of precision oncology. Nat Rev Drug Discov. 2022;21:141–62. Article CAS PubMed Google
Scholar * Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers
in 185 countries. CA Cancer J Clin. 2021;71:209–49. Article PubMed Google Scholar * Vander Heiden MG. Targeting cancer metabolism: a therapeutic window opens. Nat Rev Drug Discov.
2011;10:671–84. Article Google Scholar * Pavlova NN, Zhu J, Thompson CB. The hallmarks of cancer metabolism: still emerging. Cell Metab. 2022;34:355–77. Article CAS PubMed PubMed
Central Google Scholar * Zhou X, Tian C, Cao Y, Zhao M, Wang K. The role of serine metabolism in lung cancer: from oncogenesis to tumor treatment. Front Genet. 2022;13:1084609. Article
CAS PubMed Google Scholar * Merino Salvador M, Gomez de Cedron M, Moreno Rubio J, Falagan Martinez S, Sanchez Martinez R, Casado E, et al. Lipid metabolism and lung cancer. Crit Rev Oncol
Hematol. 2017;112:31–40. Article PubMed Google Scholar * Streich FC Jr, Lima CD. Structural and functional insights to ubiquitin-like protein conjugation. Annu Rev Biophys.
2014;43:357–79. Article CAS PubMed PubMed Central Google Scholar * Clague MJ, Urbe S, Komander D. Breaking the chains: deubiquitylating enzyme specificity begets function. Nat Rev Mol
Cell Biol. 2019;20:338–52. Article CAS PubMed Google Scholar * Lange SM, Armstrong LA, Kulathu Y. Deubiquitinases: From mechanisms to their inhibition by small molecules. Mol Cell.
2022;82:15–29. Article CAS PubMed Google Scholar * Dewson G, Eichhorn PJA, Komander D. Deubiquitinases in cancer. Nat Rev Cancer. 2023;23:842–62. Article CAS PubMed Google Scholar *
Stegmeier F, Rape M, Draviam VM, Nalepa G, Sowa ME, Ang XL, et al. Anaphase initiation is regulated by antagonistic ubiquitination and deubiquitination activities. Nature. 2007;446:876–81.
Article CAS PubMed Google Scholar * Shan J, Zhao W, Gu W. Suppression of cancer cell growth by promoting cyclin D1 degradation. Mol Cell. 2009;36:469–76. Article CAS PubMed PubMed
Central Google Scholar * Christine A, Park MK, Song SJ, Song MS. The equilibrium of tumor suppression: DUBs as active regulators of PTEN. Exp Mol Med. 2022;54:1814–21. Article CAS PubMed
PubMed Central Google Scholar * Harrigan JA, Jacq X, Martin NM, Jackson SP. Deubiquitylating enzymes and drug discovery: emerging opportunities. Nat Rev Drug Discov. 2018;17:57–78.
Article CAS PubMed Google Scholar * Han C, Yang L, Choi HH, Baddour J, Achreja A, Liu Y, et al. Amplification of USP13 drives ovarian cancer metabolism. Nat Commun. 2016;7:13525. Article
CAS PubMed PubMed Central Google Scholar * Krassikova L, Zhang B, Nagarajan D, Queiroz AL, Kacal M, Samakidis E, et al. The deubiquitinase JOSD2 is a positive regulator of glucose
metabolism. Cell Death Differ. 2021;28:1091–109. Article CAS PubMed Google Scholar * Yu S, Zang W, Qiu Y, Liao L, Zheng X. Deubiquitinase OTUB2 exacerbates the progression of colorectal
cancer by promoting PKM2 activity and glycolysis. Oncogene. 2022;41:46–56. Article CAS PubMed Google Scholar * Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg
effect: the metabolic requirements of cell proliferation. Science. 2009;324:1029–33. Article Google Scholar * Liao Y, Yang M, Wang K, Wang Y, Zhong B, Jiang N. Deubiquitinating enzyme
OTUB1 in immunity and cancer: Good player or bad actor? Cancer Lett. 2022;526:248–58. Article CAS PubMed Google Scholar * Meng Y, Hong C, Yang S, Qin Z, Yang L, Huang Y. Roles of USP9X
in cellular functions and tumorigenesis (Review). Oncol Lett. 2023;26:506. Article CAS PubMed PubMed Central Google Scholar * Spataro V, Buetti-Dinh A. POH1/Rpn11/PSMD14: a journey from
basic research in fission yeast to a prognostic marker and a druggable target in cancer cells. Brit J Cancer. 2022;127:788–99. Article CAS PubMed PubMed Central Google Scholar * Yuan
J, Zhang G, Li X, Ma Q, Cheng W, Wang W, et al. Knocking down USP39 inhibits the growth and metastasis of non-small-cell lung cancer cells through activating the p53 pathway. Int J Mol Sci.
2020;21:8949. https://doi.org/10.3390/ijms21238949. * Verma R, Aravind L, Oania R, McDonald WH, Yates JR 3rd, et al. Role of Rpn11 metalloprotease in deubiquitination and degradation by the
26S proteasome. Science. 2002;298:611–5. Article CAS PubMed Google Scholar * Han X, Ren C, Lu C, Qiao P, Yang T, Yu Z. Deubiquitination of MYC by OTUB1 contributes to HK2 mediated
glycolysis and breast tumorigenesis. Cell Death Differ. 2022;29:1864–73. Article CAS PubMed PubMed Central Google Scholar * Zheng J, Wu S, Tang M, Xi S, Wang Y, Ren J, et al. USP39
promotes hepatocellular carcinogenesis through regulating alternative splicing in cooperation with SRSF6/HNRNPC. Cell Death Di. 2023;14:670. Article CAS Google Scholar * Skowyra A, Allan
LA, Saurin AT, Clarke PR. USP9X limits mitotic checkpoint complex turnover to strengthen the spindle assembly checkpoint and guard against chromosomal instability. Cell Rep. 2018;23:852–65.
Article CAS PubMed PubMed Central Google Scholar * Nagai H, Noguchi T, Homma K, Katagiri K, Takeda K, Matsuzawa A, et al. Ubiquitin-like sequence in ASK1 plays critical roles in the
recognition and stabilization by USP9X and oxidative stress-induced cell death. Mol Cell. 2009;36:805–18. Article CAS PubMed Google Scholar * Vong QP, Cao K, Li HY, Iglesias PA, Zheng Y.
Chromosome alignment and segregation regulated by ubiquitination of survivin. Science. 2005;310:1499–504. Article CAS PubMed Google Scholar * De Ingeniis J, Ratnikov B, Richardson AD,
Scott DA, Aza-Blanc P, De SK, et al. Functional specialization in proline biosynthesis of melanoma. PLoS One. 2012;7:e45190. Article PubMed PubMed Central Google Scholar * Liu W, Hancock
CN, Fischer JW, Harman M, Phang JM. Proline biosynthesis augments tumor cell growth and aerobic glycolysis: involvement of pyridine nucleotides. Sci Rep. 2015;5:17206. Article CAS PubMed
PubMed Central Google Scholar * Hancock CN, Liu W, Alvord WG, Phang JM. Co-regulation of mitochondrial respiration by proline dehydrogenase/oxidase and succinate. Amino Acids.
2016;48:859–72. Article CAS PubMed Google Scholar * Song X, Yang W, Wu C, Han Y, Lu Y. USP9X promotes the proliferation, invasion and metastasis of liver cancer cells through regulating
the JAK2/STAT3 signaling. Oncol Lett. 2020;20:2897–905. Article CAS PubMed PubMed Central Google Scholar * Li L, Liu T, Li Y, Wu C, Luo K, Yin Y, et al. The deubiquitinase USP9X
promotes tumor cell survival and confers chemoresistance through YAP1 stabilization. Oncogene. 2018;37:2422–31. Article CAS PubMed PubMed Central Google Scholar * Wrobel L, Siddiqi FH,
Hill SM, Son SM, Karabiyik C, Kim H, et al. mTORC2 assembly is regulated by USP9X-mediated deubiquitination of RICTOR. Cell Rep. 2020;33:108564. Article CAS PubMed Google Scholar *
Bridges CR, Tan MC, Premarathne S, Nanayakkara D, Bellette B, Zencak D, et al. USP9X deubiquitylating enzyme maintains RAPTOR protein levels, mTORC1 signalling and proliferation in neural
progenitors. Sci Rep. 2017;7:391. Article PubMed PubMed Central Google Scholar * Tanner JJ, Fendt SM, Becker DF. The proline cycle as a potential cancer therapy target. Biochemistry.
2018;57:3433–44. Article CAS PubMed Google Scholar * Guo L, Cui C, Zhang K, Wang J, Wang Y, Lu Y, et al. Kindlin-2 links mechano-environment to proline synthesis and tumor growth. Nat
Commun. 2019;10:845. Article PubMed PubMed Central Google Scholar * Zhang B, Zheng A, Hydbring P, Ambroise G, Ouchida AT, Goiny M, et al. PHGDH defines a metabolic subtype in lung
adenocarcinomas with poor prognosis. Cell Rep. 2017;19:2289–303. Article CAS PubMed Google Scholar * Uphoff CC, Drexler HG. Comparative PCR analysis for detection of mycoplasma
infections in continuous cell lines. Vitr Cell Dev Biol Anim. 2002;38:79–85. Article CAS Google Scholar * Uphoff CC, Drexler HG. Detecting mycoplasma contamination in cell cultures by
polymerase chain reaction. Methods Mol Med. 2004;88:319–26. CAS PubMed Google Scholar * Wu T, Hu E, Xu S, Chen M, Guo P, Dai Z, et al. clusterProfiler 4.0: a universal enrichment tool for
interpreting omics data. Innov (Camb). 2021;2:100141. CAS Google Scholar * Kassambara A, Kosinski M, Biecek P, Fabian S. Survminer: Drawing survival curves using ‘ggplot2’ 2021 [R package
version 0.4.9. 2016. https://CRAN.R-project.org/package=survminer. * Wee S, Wiederschain D, Maira SM, Loo A, Miller C, deBeaumont R, et al. PTEN-deficient cancers depend on PIK3CB. Proc
Natl Acad Sci USA. 2008;105:13057–62. Article CAS PubMed PubMed Central Google Scholar * Wiederschain D, Wee S, Chen L, Loo A, Yang G, Huang A, et al. Single-vector inducible lentiviral
RNAi system for oncology target validation. Cell Cycle. 2009;8:498–504. Article CAS PubMed Google Scholar * Junqueira Alves C, Dariolli R, Haydak J, Kang S, Hannah T, Wiener RJ, et al.
Plexin-B2 orchestrates collective stem cell dynamics via actomyosin contractility, cytoskeletal tension and adhesion. Nat Commun. 2021;12:6019. Article CAS PubMed PubMed Central Google
Scholar * Clancy A, Heride C, Pinto-Fernandez A, Elcocks H, Kallinos A, Kayser-Bricker KJ, et al. The deubiquitylase USP9X controls ribosomal stalling. J Cell Biol. 2021;220:e202004211.
https://doi.org/10.1083/jcb.202004211. * Kampf C, Olsson I, Ryberg U, Sjostedt E. Ponten F Production of tissue microarrays, immunohistochemistry staining and digitalization within the human
protein atlas. J Vis Exp. 2012;31:3620. https://doi.org/10.3791/3620. * Kamitani T, Kito K, Nguyen HP, Yeh ET. Characterization of NEDD8, a developmentally down-regulated ubiquitin-like
protein. J Biol Chem. 1997;272:28557–62. Article CAS PubMed Google Scholar * Kochetkova E, Becirovic T, Norberg E. Extraction of metabolites from cancer cells. Methods Mol Biol.
2022;2445:329–35. Article CAS PubMed Google Scholar Download references ACKNOWLEDGEMENTS We thank Dr. Akos Vegvari for the mass spectrometric analysis of co-immunoprecipitates that was
performed at the Proteomics Biomedicum core facility, KI (https://ki.se/en/mbb/proteomics-biomedicum) and Dr. Alex Buko, Human Metabolome Technologies, for the tumor metabolome analyses. We
would like to thank Lyudmila Krassikova, Andre Lima Queiroz, Anida Hasanovic, and Divya Nagarajan for technical assistance in the early phases of the project. The graphical abstract was
created with BioRender.com. FUNDING The work was supported by the following foundations: The Swedish Research Council (2021-01787), The Swedish Cancer Society (CAN 2017/466, CAN 2017/1015,
20 0979 PjF), The Wenner-Gren Foundations (UPD2020-0104 and UPD2021-0105), Stockholm Cancer Society (no: 174093), The Lars Hiertas Memorial Foundation (FO2021-0082, FO2022-0346,
FO2023-0543), Längmanska Kulturfonden (BA21-0787, BA22-0941), O.E och Edla Johanssons Vetenskapliga Stiftelse, Stiftelsen Sigurd & Elsa Goljes Minne (LA2022-0075, LA2023-0104,
LA2024-0145), Alex och Eva Wallströms Foundation (2023-00201, 2024-00079), and Karolinska Institute KID grant. Open access funding provided by Karolinska Institute. AUTHOR INFORMATION
AUTHORS AND AFFILIATIONS * Department of Physiology and Pharmacology, Solnavägen 9, Biomedicum, Karolinska Institutet, 171 65, Stockholm, Sweden Tina Becirovic, Boxi Zhang, Erik Norberg,
Helin Vakifahmetoglu-Norberg, Vitaliy O. Kaminskyy & Elena Kochetkova * Department of Immunology, Genetics and Pathology, Rudbeck Laboratory, Uppsala University, 751 85, Uppsala, Sweden
Cecilia Lindskog Authors * Tina Becirovic View author publications You can also search for this author inPubMed Google Scholar * Boxi Zhang View author publications You can also search for
this author inPubMed Google Scholar * Cecilia Lindskog View author publications You can also search for this author inPubMed Google Scholar * Erik Norberg View author publications You can
also search for this author inPubMed Google Scholar * Helin Vakifahmetoglu-Norberg View author publications You can also search for this author inPubMed Google Scholar * Vitaliy O. Kaminskyy
View author publications You can also search for this author inPubMed Google Scholar * Elena Kochetkova View author publications You can also search for this author inPubMed Google Scholar
CONTRIBUTIONS TB and EK performed most of the in vitro experiments. VK conceived and performed co-immunoprecipitation for the LC-MS/MS analysis and subcellular fractionation. BZ performed
bioinformatics analyses. EK and VK performed the in vivo experiments. CL performed immunohistochemistry. HVN provided critical reagents. TB, EK, and EN wrote the original manuscript with
input from all authors. TB prepared the figures. All authors analyzed and interpreted data. EN and VK supervised the research. EN and HVN designed and financed the research. EK financed the
research. All authors read and approved the final paper. CORRESPONDING AUTHORS Correspondence to Vitaliy O. Kaminskyy or Elena Kochetkova. ETHICS DECLARATIONS COMPETING INTERESTS The authors
declare no competing interests. ETHICS All animal experiments were approved by the Animal Ethical Review Board Sweden (Ethical permit No. 1185-2022) and performed following the Swedish
animal welfare legislation. ADDITIONAL INFORMATION PUBLISHER’S NOTE Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURE_TABLE_LEGENDS SUPPLEMENTARY FIGURE 1 SUPPLEMENTARY TABLE 1 SUPPLEMENTARY TABLE 2 ORIGINAL WESTERN-BLOTTING RIGHTS AND PERMISSIONS OPEN ACCESS
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as
long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third
party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the
article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright
holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Becirovic, T., Zhang, B., Lindskog,
C. _et al._ Deubiquitinase USP9x regulates the proline biosynthesis pathway in non-small cell lung cancer. _Cell Death Discov._ 10, 342 (2024). https://doi.org/10.1038/s41420-024-02111-2
Download citation * Received: 16 May 2024 * Revised: 17 July 2024 * Accepted: 18 July 2024 * Published: 29 July 2024 * DOI: https://doi.org/10.1038/s41420-024-02111-2 SHARE THIS ARTICLE
Anyone you share the following link with will be able to read this content: Get shareable link Sorry, a shareable link is not currently available for this article. Copy to clipboard Provided
by the Springer Nature SharedIt content-sharing initiative







