- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:
ABSTRACT Photoreceptors detect light through a seven-helix receptor (rhodopsin) and heterotrimeric G protein (transducin) coupled to a cyclic GMP phosphodiesterase1,2. Similar pathways are
used to amplify responses to hormones, taste and smell3,4,5. The amplification of phototransduction is reduced by a fall in cytoplasmic Ca2+ (refs 6, 7, 8, 9, 10), but it is not known how
the deactivation of rhodopsin and transducin influence this response and hence the extent and duration of phosphodiesterase activity11,12,13,14. Here we investigate this by recording the
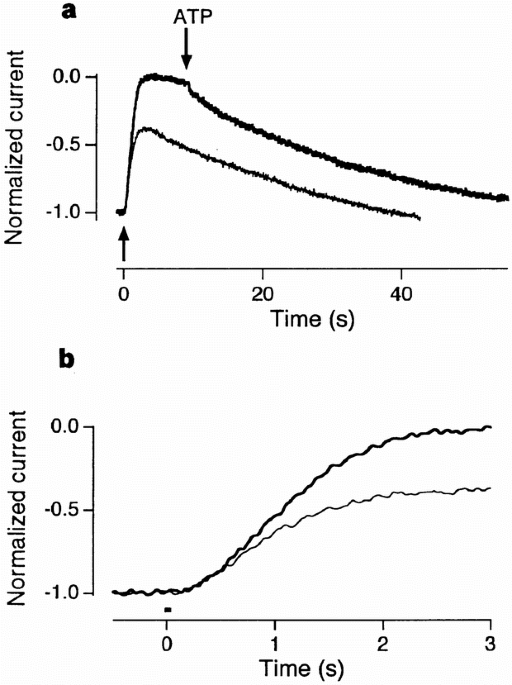
electrical response to flashes of light in truncated rod photoreceptors10. By removing ATP to block the deactivation of rhodopsin by phosphorylation15, we show that this reaction limits the
amplitude of the response and begins within 3.2 s of a flash in a solution containing 1 μM Ca2+, falling to 0.9 s in a zero-Ca2+ solution. In contrast, the activation and amplitude of the
response were unaffected when transducin deactivation by GTP hydrolysis was blocked by replacing GTP with its non-hydrolysable analogue GTP-γS11, demonstrating that there is little GTP
hydrolysis occurring over the period in which photoexcited rhodopsin is quenched. The rapid deactivation of rhodopsin is therefore a Ca2+-sensitive step controlling the amplitude of the
light response, whereas transducin deactivation is slower and controls recovery. Access through your institution Buy or subscribe This is a preview of subscription content, access via your
institution ACCESS OPTIONS Access through your institution Subscribe to this journal Receive 51 print issues and online access $199.00 per year only $3.90 per issue Learn more Buy this
article * Purchase on SpringerLink * Instant access to full article PDF Buy now Prices may be subject to local taxes which are calculated during checkout ADDITIONAL ACCESS OPTIONS: * Log in
* Learn about institutional subscriptions * Read our FAQs * Contact customer support SIMILAR CONTENT BEING VIEWED BY OTHERS FUNCTIONAL MODULATION OF PHOSPHODIESTERASE-6 BY CALCIUM IN MOUSE
ROD PHOTORECEPTORS Article Open access 26 April 2021 ULTRAFAST STRUCTURAL CHANGES DIRECT THE FIRST MOLECULAR EVENTS OF VISION Article Open access 22 March 2023 ACTIVE STATE STRUCTURES OF A
BISTABLE VISUAL OPSIN BOUND TO G PROTEINS Article Open access 16 October 2024 REFERENCES * Stryer, L. The cyclic nucleotide cascade of vision. _Annu. Rev. Neurosci._ 9, 87–119 (1986).
Article CAS Google Scholar * Lagnado, L. & Baylor, D. Signal flow in visual transduction. _Neuron_ 8, 995–1002 (1992). Article CAS Google Scholar * Lamb, T. D. & Pugh, E. N.
G-protein cascades: gain and kinetics. _Trends Neurosci._ 15, 291–298 (1992). Article CAS Google Scholar * Kolesnikov, S. S. & Margolskee, R. F. Acyclic-nucleotide-suppressible
conductance activated by transducin in taste cells. _Nature_ 376, 85–88 (1995). Article ADS CAS Google Scholar * Gold, G. H. & Pugh, E. N. The nose leads the eye. _Nature_ 385,
677–679 (1997). Article ADS CAS Google Scholar * Matthews, H. R., Murphy, R. L., Fain, G. L. & Lamb, T. D. Photoreceptor light adaptation is mediated by cytoplasmic calcium
concentration. _Nature_ 334, 67–69 (1988). Article ADS CAS Google Scholar * Nakatani, K. & Yau, K.-W. Calcium and light adaptation in retinal rods and cones. _Nature_ 334, 69–71
(1988). Article ADS CAS Google Scholar * Kawamura, S. & Murakami, M. Calcium-dependent regulation of cyclic GMP phosphodiesterase by a protein from frog retinal rods. _Nature_ 349,
420–423 (1991). Article ADS CAS Google Scholar * Kawamura, S. Rhodopsin phosphorylation as a mechanism of cyclic GMP phosphodiesterase regulation by S-modulin. _Nature_ 362, 855–857
(1993). Article ADS CAS Google Scholar * Lagnado, L. & Baylor, D. A. Calcium controls light-triggered formation of catalytically active rhodopsin. _Nature_ 367, 273–277 (1994).
Article ADS CAS Google Scholar * Vuong, T. M. & Chabre, M. Deactivation kinetics of the transduction cascade of vision. _Proc. Natl Acad. Sci. USA_ 88, 9813–9817 (1991). Article ADS
CAS Google Scholar * Pepperberg, D. R. et al. Light-dependent delay in the falling phase of the retinal rod photoresponse. _Vis. Neurosci._ 8, 9–18 (1992). Article CAS Google Scholar
* Lyubarsky, A., Nikonov, S. & Pugh, E. N. The kinetics of inactivaction of the rod phototransduction cascade with constant Ca12+. _J. Gen. Physiol._ 107, 19–34 (1996). Article CAS
Google Scholar * Murnick, J. G. & Lamb, T. D. Kinetics of desensitisation induced by saturating flashes in toad and salamander rods. _J. Physiol. (Lond.)_ 495, 1–13 (1996). Article CAS
Google Scholar * Wilden, U., Hall, S. W. & Kuhn, H. Phosphodiesterase activation by photoexcited rhodopsin is quenched when rhodopsin is phosphorylated and binds the intrinsic 48 kD
protein of rod outer segments. _Proc. Natl Acad. Sci. USA_ 83, 1174–1178 (1986). Article ADS CAS Google Scholar * Detwiler, P. B. & Gray-Keller, M. P. The mechanisms of vertebrate
light adaptation: speeded recovery versus slowed activation. _Curr. Opin. Neurobiol._ 6, 440–444 (1996). Article CAS Google Scholar * Chen, J., Makino, C. L., Peachey, N. S., Baylor, D.
A. & Simon, M. I. Mechanisms of rhodopsin inactivation _in vivo_ as revealed by a COOH-terminal truncation mutant. _Science_ 267, 374–377 (1995). Article ADS CAS Google Scholar *
Pepperberg, D. R., Jin, J. & Jones, G. J. Modulation of transduction gain in light adaptation of retinal rods. _Vis. Neurosci._ 11, 53–62 (1994). Article CAS Google Scholar * Corson,
D. W., Cornwall, M. C. & Pepperberg, D. R. Evidence for the prolonged photoactivated lifetime of an analogue visual pigment containing 11-_cis_ 9-desmethylretinal. _Vis. Neurosci._ 11,
91–98 (1994). Article CAS Google Scholar * Nikonov, S. S., Lyubarsky, A. L. & Pugh, E. N. Hydroylamine decreases rod sensitivity _in situ_ without changing the dominant time constant
of recovery. _Invest. Ophthalmol. Vis. Sci._ 37, S5 (1996). Google Scholar * McNaughton, P. A., Cervetto, L. & Nunn, B. J. Measurement of the intracellular free calcium concentration in
salamander rods. _Nature_ 322, 261–263 (1986). Article ADS CAS Google Scholar * Matthews, H. R. Static and dynamic actions of cytoplasmic Ca2+ in the adaptation of responses to
saturating flashes in salamander rods. _J. Physiol. (Lond.)_ 490, 1–15 (1996). Article CAS Google Scholar * Lagnado, L., Cervetto, L. & McNaughton, P. A. Calcium homeostasis in the
outer segments of retinal rods from the tiger salamander. _J. Physiol. (Lond.)_ 455, 111–142 (1992). Article CAS Google Scholar * Gray-Keller, M. P. & Detwiler, P. B. The calcium
feedback signal in the phototransduction cascade of vertebrate rods. _Neuron_ 13, 849–861 (1994). Article CAS Google Scholar * Cervetto, L., Lagnado, L., Perry, R. J., Robinson, D. W.
& McNaughton, P. A. Extrusion of calcium from rod outer segments is driven by both sodium and potassium gradients. _Nature_ 337, 740–743 (1989). Article ADS CAS Google Scholar *
Younger, J. P., McCarthy, S. T. & Owen, W. G. Modulation of the cytosolic free calcium and changes in sensitivity and circulating current occur over the same range of steady-state
adapting lights in rod photoreceptors. _Invest. Ophthalmol. Vis. Sci._ 33, 1104 (1992). Google Scholar * Tanaka, T., Ames, J. B., Harvey, T. S., Stryer, L. & Ikura, M. Sequestration of
the membrane-targeting myristoyl group of recoverin in the calcium-free state. _Nature_ 376, 444–447 (1995). Article ADS CAS Google Scholar * Jones, G. J. Light adaptation and the rising
phase of the flash photocurrent of salamander retinal rods. _J. Physiol. (Lond.)_ 487, 441–451 (1995). Article ADS CAS Google Scholar * Gray-Keller, M. P. & Detwiler, P. B. Ca2+
dependence of dark- and light-adapted flash resonses in rod photoreceptors. _Neuron_ 17, 323–331 (1996). Article CAS Google Scholar * Sagoo, M. S. & Lagnado, L. Ca2+ increases the
apparent affinity of transducin for GTP. _Invest. Ophthalmol. Vis. Sci._ 38, S614 (1997). Google Scholar Download references ACKNOWLEDGEMENTS We thank C. Raeburn of the LMB Instrumentation
Workshop for assistance. This work was supported by the HFSP. M.S.S. held a scholarship of the University of Cambridge M.B, Ph.D. Programme. AUTHOR INFORMATION AUTHORS AND AFFILIATIONS * MRC
Laboratory of Molecular Biology, Hills Road, CB2 2QH, Cambridge, UK Mandeep S. Sagoo * MRC Laboratory of Molecular Biology, Hills Road, CB2 2QH, Cambridge, UK Leon Lagnado Authors * Mandeep
S. Sagoo View author publications You can also search for this author inPubMed Google Scholar * Leon Lagnado View author publications You can also search for this author inPubMed Google
Scholar CORRESPONDING AUTHOR Correspondence to Leon Lagnado. RIGHTS AND PERMISSIONS Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Sagoo, M., Lagnado, L. G-protein
deactivation is rate-limiting for shut-off of the phototransduction cascade. _Nature_ 389, 392–395 (1997). https://doi.org/10.1038/38750 Download citation * Received: 01 May 1997 * Accepted:
07 July 1997 * Issue Date: 25 September 1997 * DOI: https://doi.org/10.1038/38750 SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get
shareable link Sorry, a shareable link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt content-sharing initiative

:max_bytes(150000):strip_icc():focal(216x0:218x2)/benedict-cumberbatch-1-435-4-20cc736017b24435a3498a49d7c22b0e.jpg)